Electrical conductivity meter with matched conductivity electrodes provides a measuring range from 0.02μs/cm~0.02ms/cm to 0.02ms/cm~20ms/cm, L/H alarm, analog output 4-20mA and RS485 communication function. Digital conductivity meter can be used for online conductivity monitoring of different liquids such as pure water, tap water, sewage, and concentrated liquid.

Digital conductivity meter is compatible with pure water/tap water/sewage.
- 2.4 inches 12864 lattice screen.
- Isolating transmitting output, with little interference.
- Digital conductivity meter provides EC/TDS/ER measurement, temperature measurement, top/bottom limit control, transmitting output, 4-20mA analog output, and RS485 communication.
- The conductivity tester has configurable manual and auto temperature offset functions, upper/lower limit warning and delay, hummer and LCD backlight switch.
- Industrial controlled door keep, to avoid instrument halted.
Applications
sisco conductivity meter, an intelligent online chemical analyzer, is widely applied for continuous monitoring and measurement of EC value or TDS value or ER value and temperature in the solution in the industry of thermal power, chemical fertilizer, environmental protection, metallurgy, pharmacy, biochemistry, food and water, etc.

| Model | SISCO-CDTYM-TDS210-B |
| Shipping weight | 1.5kg |
| Dimension | 96*96*132mm (H*W*D), with hole size 92.5*92.5mm |
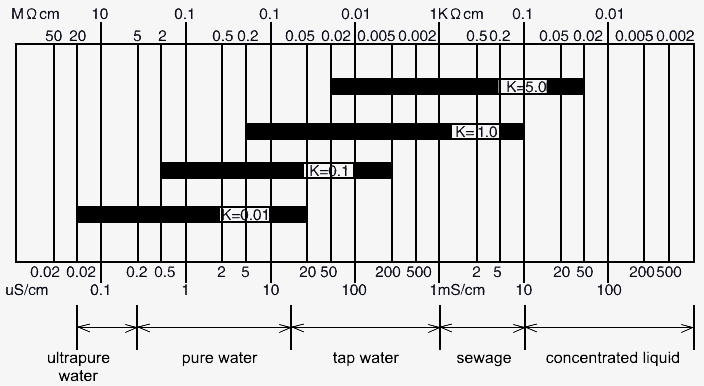
| Measuring range * | Conductivity electrode (K=0.01): 1.00~20.00μs/cm Conductivity electrode (K=0.1): 1.0~200.0μs/cm Conductivity electrode (K=1.0): 2~2000μs/cm Conductivity electrode (K=10.0): 20~20000μs/cm (0.02~20ms/cm) |
| Accuracy | ±1%FS |
| Stability | ±1%FS/24h |
| Temperature compensation | -10~+130℃ (14~266℉) manual/automatic |
| Electrode built-in sensor | PT1000, accuracy: ±0.5℃ |
| Display | 2.4" LCD display |
| Output signal | 2 relay outputs with capacity 250VAC/3A 4-20mA analog output RS485 output with standard MODBUS-RTU |
| Power supply | 220VAC±10%, 50/60Hz |
Conductivity electrode dimension
| Electrode constant | Electrode material | Length below thread | Diameter | Diameter of measuring the hole | Thread | Lead wire length |
| K=0.01 | 316 stainless steel | 63mm | 13mm | 6mm | G3/4 | 10m |
| K=0.1 | 316 stainless steel | 61mm | 13mm | 6mm | G3/4 | |
| K=1.0 | 316 stainless steel | 61mm | 13mm | 6mm | G3/4 | |
| K=10.0 | Polysulfone | 69.5mm | 23.3mm | 6mm | 3/4NPT |
Reference diagram for electrode selection
Q1: What is a conductivity meter?
The conductivity meter is mainly an instrument for measuring the current capacity of the liquid medium. They are generally used for continuous monitoring of conductivity values in electric power, the chemical industry, metallurgy, environmental protection, pharmaceuticals, fields, lakes, scientific research, food and tap water, as well as in water treatment and aquaculture experiments.
Q2: How does a conductivity meter work?
Conductivity Meter Principle Conductivity is a numerical representation of the ability of a solution to conduct electrical current. The conductivity of water has a certain relationship with the number of inorganic acids, alkalis and salts contained in the water. When their concentrations are low, the conductivity increases with the increase of the concentration. Therefore, this indicator is often used to infer the total concentration or salinity of ions in water. Conductance (G) is the inverse of resistance (R). As a result, when two electrodes (usually platinum or platinum black) are inserted into the solution, the resistance R between the two electrodes can be measured. According to Ohm's law, the resistance value is proportional to the electrode spacing L (cm) and inversely proportional to the electrode's cross-sectional area a (cm2 ).
Q3: What is a conductivity meter used for?
- Water quality monitoring: The conductivity is affected by salt, inorganic acid, alkali, etc. in the water, so the higher the conductivity, the higher the concentration in the water will be reflected, the total concentration and salt content of ions in the water can be understood, and the purity of the water quality and due to the control of the production process, the conductivity of the water will vary with the TDS content in the water.
- Electroplating solution: Conductivity is an important parameter reflecting the electroplating solution. It can reduce the voltage in the electroplating bath and improve the dispersion ability of the electroplating solution. The conductivity of the electroplating solution can be reflected by measuring the conductivity.
- Atmospheric monitoring: The conductivity meter is used in atmospheric monitoring mainly for pollutants contained in the atmosphere such as carbon monoxide, sulfur dioxide, carbon dioxide and various nitrogen oxides. It can pass the gas absorption device and pass the gas through the absorption liquid to check the change of conductivity and monitor exceeding the standard in the polluted area
Tips: How to determine the conductance cell constant?
According to the formula K = S / G, the electrode constant K can be obtained by measuring the conductivity G of the conductivity electrode in a certain concentration of KCL solution. At this time, the conductivity S of the KCL solution is known. Because the concentration and temperature of the measuring solution are different, and the accuracy and frequency of the conductivity meter are also different, a large error sometimes occurs in the conductivity electrode constant K. And the electrode constant may also change after a period of use. Therefore, the electrode constants of newly purchased conductivity electrodes and conductivity electrodes after a period of use should be re-calibrated.
Pay attention to the following points when measuring the conductivity constant of the conductivity electrode:
- The supporting conductivity meter should be used for measurement, and don't use other types of conductivity meters.
- The temperature of the KCL solution should be preferably close to the temperature of the actually measured solution.
- The concentration of the KCL solution should be preferably near the concentration of the actually measured solution.
Thank you for buying industrial test and measurement equipment on SISCO.com, all products sold by SISCO and the partner cover a 12 months warranty, effective from the date of receiving the products.
What is covered?
SISCO is responsible for providing free spare parts, and free technical support to assist the customer to repair the defective products until the problem is solved.
What is not covered?
- Product purchased from anyone other than a SISCO store or a SISCO authorized reseller.
- Expendable parts.
- Routine cleaning or normal cosmetic and mechanical wear.
- Damage from misuse, abuse or neglect.
- Damage from use of parts other than SISCO approved.
- Damage from use outside the product’s usage or storage parameters.
- Damage from use of parts not sold by SISCO.
- Damage from modification or incorporation into other products.
- Damage from repair or replacement of warranted parts by a service provider other than a SISCO authorized service provider.
- Damage caused by the application environment not meeting the product usage requirements and the failure to perform preventive maintenance.

