A salinometer is a valuable tool for measuring the salt concentration in various solutions, especially in oceanography, aquaculture, and water quality monitoring. To ensure accurate and reliable salinity measurements, periodic calibration is essential. In this blog, we will guide you through the process of calibrating a salinometer, covering calibration standards and different calibration methods.
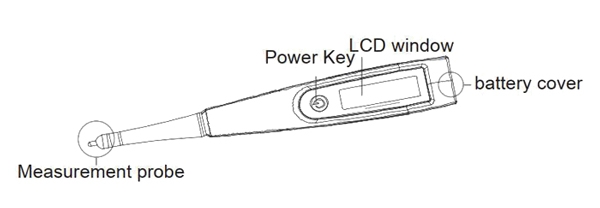
Salinometer Basics
Salinity is usually expressed in parts per thousand (ppt) or practical salinity units (PSU), with 1 ppt roughly equivalent to 1 PSU. Salinometers work by measuring the electrical conductivity or other properties of a liquid to estimate the concentration of dissolved salts. These measurements can be important for understanding water quality, assessing the health of aquatic ecosystems, and ensuring the proper functioning of certain industrial processes where salt levels need to be controlled.
Calibration Standards for Salinometers
Salinometers primarily utilize the principle of conductivity cells to determine the salinity of a sample by measuring its conductivity relative to a standard solution of seawater. The calibration procedure involves inspection of the device's appearance, checking for salinity indication errors, and evaluating salinity repeatability.
The calibration standards used for salinometer calibration include:
- Preparation of seawater standards: Precise dilution using the weight-volume method is required to prepare standard solutions with salinity values uniformly distributed across the salinity range.
- Temperature control is crucial for ensuring measurement accuracy. Temperature has a significant impact on salinity, with higher temperatures corresponding to higher salinity values. Maintaining consistent temperature control ensures the accuracy of salinity values. Temperature stability refers to both instrument temperature and solution temperature.
Salinometer calibration is typically conducted at 15°C and standard atmospheric pressure, using the ratio of the conductivity of the tested solution to a standard solution of potassium chloride with a mass ratio of 32.4356 × 10^-3. When the ratio (K) precisely equals 1, the salinity value is 35.000. This salinity standard value is essentially a reference for potassium chloride conductivity and is dimensionless, making it unsuitable for the calibration of non-seawater salinometers.
For salinometers, there is no specific set of items to calibrate or reference standards to consider, particularly for salinometers that only display salinity values. It is often impossible to calibrate the electronic components of these instruments. Salinometer calibration is not applicable to metrology for oceanographic salinometers and has certain limitations for non-oceanographic salinometers.
Calibration Methods for Salinometers
Oceanographic salinometers operate based on principles including density, optics, and conductivity. Currently, only salinometers employing the principle of conductivity have corresponding metrological procedures that can be referenced.
For oceanographic salinometers utilizing the density principle, seawater hydrometers can be selected as reference instruments. Each set of seawater hydrometers typically includes one broad-range hydrometer and seven precision hydrometers. The salinity at a specific temperature can be determined by measuring the position of the meniscus projection on the hydrometer scale and referring to a temperature-salinity table. Performance evaluation of the instrument can be carried out based on indication errors and repeatability.
For oceanographic salinometers using the optical principle, standard seawater materials can also be employed. Calibration typically involves following the manufacturer's instructions, then uniformly adding standard seawater to the prism, eliminating air bubbles, and conducting analytical readings. During the experiment, temperature control is essential to ensure equilibrium between the prism temperature and the solution temperature. Furthermore, since optical systems are used, measurements should be taken in the direction of light and not against it to avoid causing reading deviations.
Non-oceanographic salinometers primarily operate based on optical and conductivity principles. At present, metrological traceability for non-oceanographic salinometers is relatively limited. Through research and analysis of non-oceanographic salinometers available in the market, it has been observed that most of these devices use ppt, ppm, or % as display units, with measurement ranges mostly within 0-10%, and measurement results are often expressed as mass percentages of sodium chloride.
Moreover, temperature has a significant influence during the testing process for such salinometers. Generally, higher temperatures correspond to higher salinity values. Many non-oceanographic salinometers in the market are handheld portable devices without temperature control baths. Although some instruments are designed with temperature compensation in mind, special attention should be given to temperature effects to ensure consistency between solution temperature and instrument temperature. Additionally, conductivity-based salinometers often employ highly sensitive electrode components. Therefore, during measurements, it is essential to ensure thorough contact between the solution and the electrode, allowing for a stable period to ensure stable and accurate readings.
The salinity meter is an important environmental measurement tool. Making good use of it will greatly improve the efficiency of measurement work.

